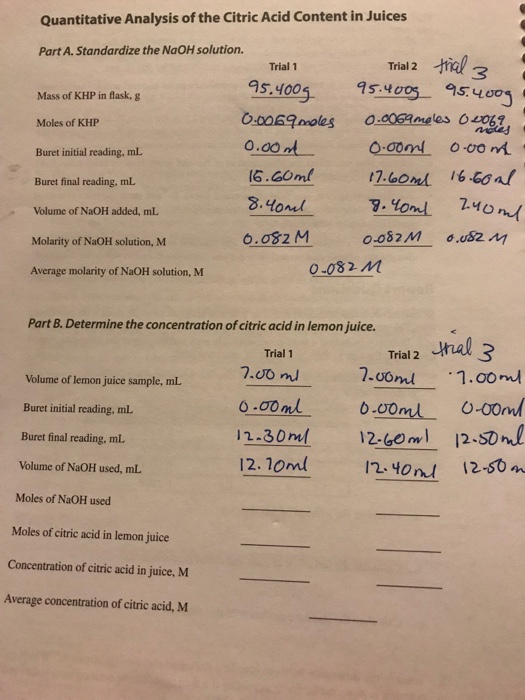
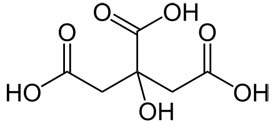
The Case for Bottled Lemon Juice in Canning • AnswerLine • Iowa State University Extension and Outreach

PDF) Quantitative Assessment of Citric Acid in Lemon Juice, Lime Juice, and Commercially-Available Fruit Juice Products

Serum with Vitamin C or Essential Citrus Oil. Lemon Juice Citric Acid. Concept Stock Photo - Image of blue, flatlay: 136663716