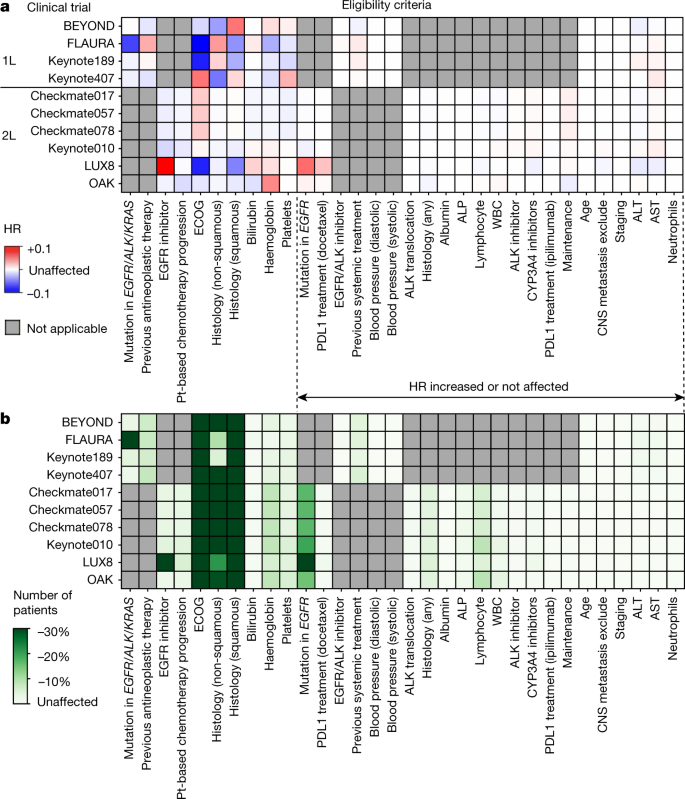
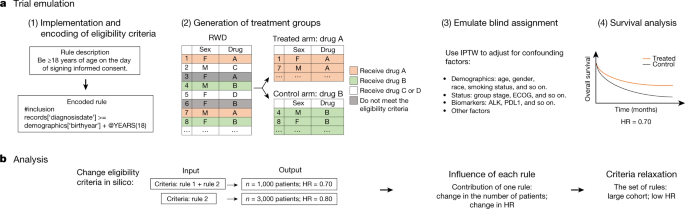
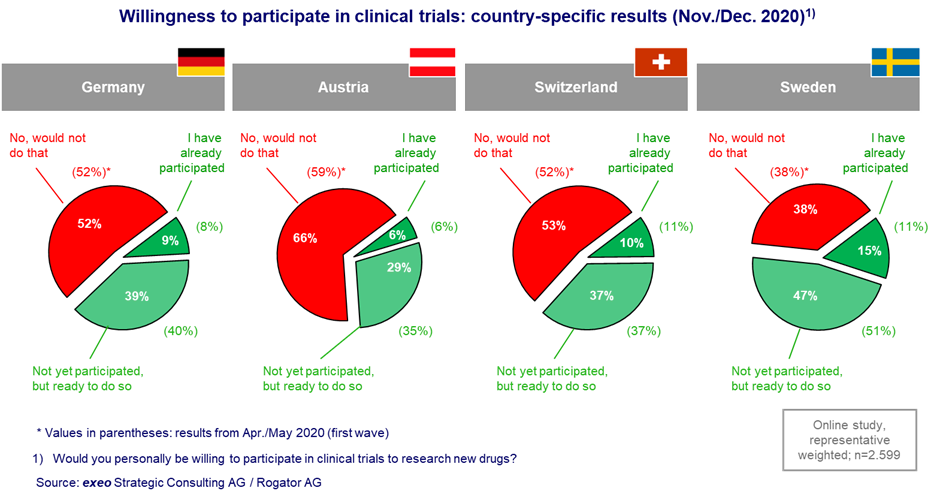
Interpreting Clinical Trial Results Betty Anne Schwarz MSc, BA, RN OHRI Clinical Research Conference Workshop October 16, ppt download

Accessibility of trial reports for drugs stalling in development: a systematic assessment of registered trials – topic of research paper in Clinical medicine. Download scholarly article PDF and read for free on

Generalizability of findings from randomized controlled trials is limited in the leading general medical journals - Journal of Clinical Epidemiology

Evidence-informed recommendations to reduce dissemination bias in clinical research: conclusions from the OPEN (Overcome failure to Publish nEgative fiNdings) project based on an international consensus meeting – topic of research paper in

Barriers and enablers to locally-led clinical trial conduct in low and middle income countries: strategies for developing locally sustainable health research capacity | Semantic Scholar